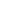Fenbufen
Indications: antipyretic and analgesic, nonsteroidal anti-inflammatory drug
Classification:
Detailed
| Molecular formula | C₁₆H₁₄O₃ |
| Molecular weight | 254.29 |
| Structural formula |

|
| Storage | Store in a dark, airtight container. |
| Validity period | 48 months |
| Implementation Standard | Volume II of the 2025 Edition of the Pharmacopoeia of the People’s Republic of China |
| Appearance | White or off-white crystalline powder; odorless. |
| Identification | Compliant with regulations |
| Melting point | 185–188°C |
| Chloride | Compliant with regulations |
| Sulfate | Compliant with regulations |
| Related substances | Compliant with regulations |
| Loss on drying | ≤0.5% |
| Residue on ignition | ≤0.1% |
| Heavy metals | ≤10PPM |
| Arsenic salt | ≤0.001% |
| Content (on a dry basis) | ≥98.5% |
Key words:
Raw material products
fenbufen
Previous Page:
Next page:
Related Products
Online Inquiry